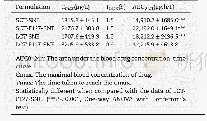
《Table 2–Pharmacokinetic parameters of formulations.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《The accelerated blood clearance phenomenon of PEGylated nanoemulsion upon cross administration with nanoemulsions modified with polyglycerin》
aAREA under curve from time 0 to 60 min.bThe mean retention time from time 0 to 60 min.
For evaluating the effect of PG610-DSPE on the induction of the ABC phenomenon in rats,rats were treated with the 1%GE-B,3%GE-B,9%GE-B and 9%PE-B at 5μmol phospholipid/kg as the first dose respectively.7 d later,the rats were injected with the9%PE(5μmol phospholipid/kg).The result of Fig.3A showed that,the plasma concentrations of the second dose of the 9%PEdecrease.Compared with control group which pretreated with5%glucose,rats pretreated with the 1%GE-B induce the rapidly clearance of the 9%PE(AUC (0–60 min)values,5723.06±85.14 and1236.65±27.23μg/ml/min,respectively,**P<0.01,Table 2) .The decreasing extends from strong to weak are 1%GE>3%GE>9%GE(MRT (0–60)(min),19.45±1.54,22.48±2.12,23.49±2.76 min respectively) .This suggests that the ABC phenomenon is induced by the nanoemulsion coated with PG610-DSPE.This is the first report that the GE induces the ABC phenomenon of the second dose of the 9%PE.As shown in the Table 2,pharmacokinetic parameters reveal that rats pretreated with the 9%PE-B can induce stronger ABC phenomenon than the 1%GE-B(AUC (0–60 min)values,204.15±9.38 and 1236.65±27.23μg/ml/min,respectively) .The parameters between the control group and the group pretreated with the 1%GE-B are compared.The differences in parameters between these two groups are significant(**P<0.01)(Table 2) .The Fig.2B shows that the liver and spleen accumulations are increased due to the induction of the ABCphenomenon.As we all know,PEG endows the nanoparticles“stealth”.Some researchers think that the reason why the PEGylated nanoemulsion induces the ABC phenomenon is that the long lifetime of nanoparticles trigger the immune system[40].However,from the results of our work,the ABC phenomenon is also induced by the nanocarriers in short circulation.
| 图表编号 | XD00186254100 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.01.01 |
| 作者 | Yuqing Su、Lirong Wang、Kaifan Liang、Mengyang Liu、Xinrong Liu、Yanzhi Song、Yihui Deng |
| 绘制单位 | Shenyang Pharmaceutical University、Shenyang Pharmaceutical University、Shenyang Pharmaceutical University、Shenyang Pharmaceutical University、Shenyang Pharmaceutical University、Shenyang Pharmaceutical University、Shenyang Pharmaceutical University |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 2–Pharmacokinetic parameters of formulations.”的人还看了
-
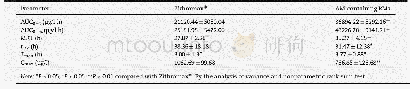
- Table 5–Pharmacokinetic parameters of azithromycin in rats after oral administration of AM-containing RMs and Zithromax.
-

- Table 1–The pharmacokinetic parameters of TMP in joint tissue and synovial fluid after intra-articular injection of TMP
-

- Table 3–The main pharmacokinetic parameters of CTN after oral administration of the optimized CTN-SRPs in rats (n=6) in