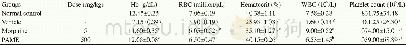《Table 5Pharmacokinetic parameters of GRe after vaginal administration (mean±SD, n=5) .》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Pharmacokinetic study of ginsenoside Re after vaginal administration in rabbits by UPLC-MS/MS determination》
The quantification determination was in the selected ion monitoring(SIM)mode using an electrospray negative ionization(ESI-).The ESI condition was optimized as follows:gas temperature350°C;gas flow rate 11 L/min;nebulizer 15 psi;and capillary4000 V(Agilent Technologies,USA).The concrete quantitative parameters were listed in Table 1.The Agilent Mass Hunter Workstation was used for data acquisition and peak integration.
| 图表编号 | XD008001000 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.07.18 |
| 作者 | Xu Han、Ying Zhang、Jia-xin Pi、Dereje Kebebe、Bing Zhang、Shu-ya Wang、Bo-ying Liu、Jing Ren、Zhi-dong Liu |
| 绘制单位 | Tianjin State Key Laboratory of Modern Chinese Medicine,Tianjin University of Traditional Chinese Medicine、Engineering Research Center of Modern Chinese Medicine Discovery and Preparation Technique,Ministry of Education,Tianjin University of Traditional C |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 5Pharmacokinetic parameters of GRe after vaginal administration (mean±SD, n=5) .”的人还看了
-
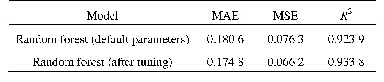
- Table 1 Comparison of fitting effects of random forest regression model before and after parameter tuning
-
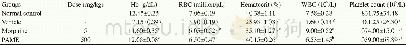
- Table 1Influence of PAME on hematological parameters after vincristine induced neuropathic pain in rats.
-
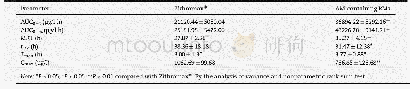
- Table 5–Pharmacokinetic parameters of azithromycin in rats after oral administration of AM-containing RMs and Zithromax.
-

- Table 1–The pharmacokinetic parameters of TMP in joint tissue and synovial fluid after intra-articular injection of TMP