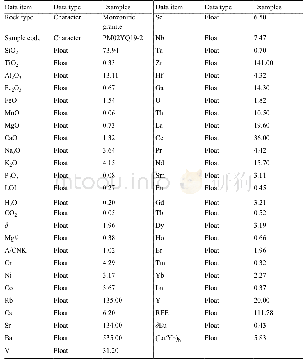
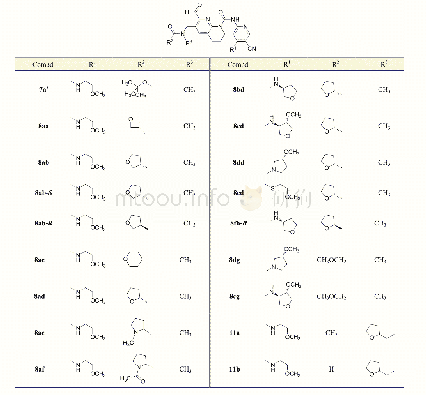
《Table 1Effect of the structure of ZTQAs.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《基于两性离子型季铵盐-KI的温度控制自分离离子液体(IL)催化体系用于二氧化碳固定反应(英文)》
Reaction conditions:PO 50 mmol,ZTQAs 1.0 mol%,KI 1.0 mol%,120°C,1.0 MPa,5 h.a No reaction.The selectivity to products is>99%for all.
Generally,the polarity of molecules can be modified by changing the length of the alkyl chain,which has a profound impact on catalysis.Therefore,a series of ZTQAs with varying alkyl chain lengths was synthesized(Scheme 1).The effect of various ZTQAs with KI on the coupling reaction of CO2 was investigated.The results are illustrated in Table 1,with the model reaction of PO and CO2.Nearly no reaction was observed with ZTQAs as the only catalyst,whereas KI could catalyze the coupling reaction alone,although the yield was relatively low(entry 1).Notably,when both ZTQAs and KI were in the reac‐tion,an increase in the yield of propylene carbonate(PC)wasdetected(entries 2–7).Thus,it is clear that ZTQAs play an im‐portant role in realizing the coupling reaction.In addition,ZTQAs with varying alkyl chain lengths exhibited different cat‐alytic performances,even though similar catalytic activity was observed for the alkyl chain group ranging from butyl to tetradecyl,as demonstrated(entries 2–5).Unfortunately,by prolonging the length of the alkyl chain further,an opposite tendency was observed(entries 6,7).When the catalyst was DTPS with tetradecyl,the best yield obtained was 75.5%.To investigate this,the solubility of various ZTQAs in PC was ana‐lyzed and listed in Fig.S1 and S2.At the initial stage of heating,all the catalytic systems,except for those of DHPS/KI and DSPS/KI,dissolved completely in PC,resulting in lower activi‐ties for these two systems.After cooling,both DDPS/KI and DTPS/KIprecipitatedspontaneously.Thetempera‐ture‐controlled self‐separation behavior that can be achieved by modifying the length of the alkyl chain(Scheme 2)provided a convenient method to reuse catalysts.Meanwhile,the tem‐perature‐controlled phenomenon was observed in the reaction process for the catalytic system and listed in Fig.S3.Further‐more,the system of DTPS/KI was further investigated in detail.
| 图表编号 | XD008199800 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.11.01 |
| 作者 | 付西英、谢鹏涛、连一苇、何乐芹、赵伟、常涛、秦身钧 |
| 绘制单位 | 河北工程大学材料科学与工程学院、邯郸学院化学化工与材料学院、河北工程大学材料科学与工程学院、河北工程大学材料科学与工程学院、南阳师范学院化学与制药工程学院、河北工程大学材料科学与工程学院、河北工程大学材料科学与工程学院 |
| 更多格式 | 高清、无水印(增值服务) |