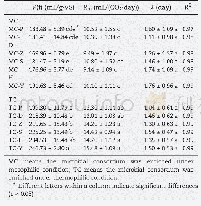
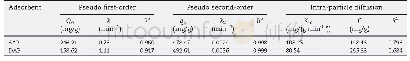
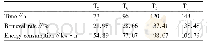《Table 1 Macroscopic kinetic parameters of toluene consumption》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Kinetics and Mechanism Modeling of Liquid-Phase Toluene Oxidation to Benzaldehyde Catalyzed by Mn–Mo Oxide》
Figure 1 shows that the natural logarithms of the measured conversion rate of toluene have a linear relationship with residence time over the interval of 413–443 K.This shows that the oxidation rate of toluene obeys fi rst-order principles within the scope of these experimental conditions.The kinetic parameters are enumerated in the fi rst two columns in Table 1 and the macroscopic oxidation of toluene can be embodied into Eq.(2):
| 图表编号 | XD0042636600 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.02.01 |
| 作者 | Wang Li、Qingjun Zhang、Aiwu Zeng |
| 绘制单位 | School of Chemical Engineering and Technology, Tianjin University、School of Chemical Engineering and Technology, Tianjin University、School of Chemical Engineering and Technology, Tianjin University |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1 Macroscopic kinetic parameters of toluene consumption”的人还看了
-

- Table 5 Eff ect of Mn/Mo ratio on macroscopic kinetic parameters of benzaldehyde generation from toluene