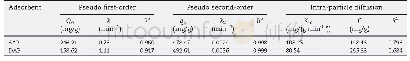
《Table 1–Kinetics parameters of MB adsorption onto single network alginate/polyvinyl alcohol beads (
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Dye removal by eco-friendly physically cross-linked double network polymer hydrogel beads and their functionalized composites》
The influence of contact time on adsorption capacity is shown in Fig.1c.It can be seen that the adsorption capacity increases sharply within 3 hr,slowly increases thereafter finally reaches equilibrium around 10 hr.The adsorption equilibrium time of the SAP is longer than that of the DAP Based on these results,10 hr is taken as the equilibrium time in adsorption experiments.Furthermore,the kinetic data are fitted by three models.The linear plots are shown in Fig.1d–f.All calculated model parameters are shown in Table 1.
| 图表编号 | XD0033524600 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.04.15 |
| 作者 | Yan Kong、Yuan Zhuang、Zhiyong Han、Jianwei Yu、Baoyou Shi、Kun Han、Haotian Hao |
| 绘制单位 | Key Laboratory of Drinking Water Science and Technology,Research Center for Eco-Environmental Sciences,Chinese Academy of Sciences、College of Petrochemical Engineering,Lanzhou University of Technology、Key Laboratory of Drinking Water Science and Technolog |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1–Kinetics parameters of MB adsorption onto single network alginate/polyvinyl alcohol beads (SAP) and dual network”的人还看了
-
- Table 4 Adsorption kinetics parameters for Pseudo-first-order model and Pseudosecond-order model of Ni2+on HBCx
-

- 表2 对COD和氮、磷的吸附动力学拟合参数Tab.2 Fitting parameters of adsorption kinetics of COD, N and P in wastewater by adding materials





