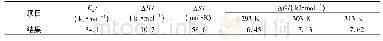《Table 2–Isotherm parameters for HA adsorption onto SPAC-NH2.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Efficient capture of aqueous humic acid using a functionalized stereoscopic porous activated carbon based on poly(acrylic acid)/food-waste hydrogel》
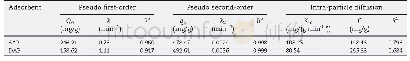
The two kinetics models are shown in Fig.4b.The results of the kinetic constants and correlation coefficients(R2)are listed in Table 4.A higher correlation coefficient(R2=0.996)was achieved for the pseudo-first-order model,which showed that the adsorption of HA onto SPAC-NH2was fit better by the pseudo-first-order model.
| 图表编号 | XD0033517000 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.03.15 |
| 作者 | Tao Zhou、Xin Zhao、Shuya Wu、Lianghu Su、Youcai Zhao |
| 绘制单位 | The State Key Laboratory of Pollution Control and Resource Reuse, School of Environmental Science and Engineering, Tongji University、Shanghai Chengpei Enterprise Management Consulting Co., Ltd.、The State Key Laboratory of Pollution Control and Resource Re |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 2–Isotherm parameters for HA adsorption onto SPAC-NH2.”的人还看了
-
- Table 3–Langmuir and Freundlich adsorption isotherm for TMA adsorption by coir pith, lignin and holocellulose extracted
-

- 表1 PEI改性磁性四氧化三铁对水中刚果红等温吸附模型参数Tab.1 Isotherm parameters for Congo red adsorption onto surfactant-modified magnetite Fe3O4