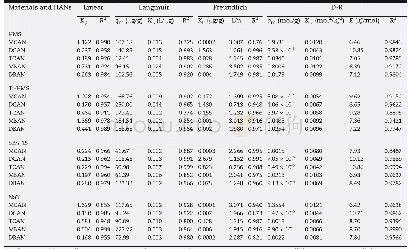
《Table 1–Langmuir, Freundlich and Dubinin–Radushkevich isotherm parameters for the adsorption of cop
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Facile one-step synthesis of functionalized biochar from sustainable prolifera-green-tide source for enhanced adsorption of copper ions》
The effects of additive agents on the pore structure and adsorption ability were studied by a series of experiments at the hydrothermal temperature of 140°C,hydrothermal time of8 hr and impregnation ratio of 1.Appendix A Table S1 shows that the biochars obtained by hydrothermal carbonization with ammonium phosphate,thiocarbamide,ammonium chloride and urea displayed specific surface areas of 6.025,326.5,6.781 and 2.612 m2/g,respectively.Biochars produced with thiocarbamide exhibited greater surface area than those produced with the other additives.Nevertheless,it is important to emphasize that the biochars produced with thiocarbamide did not show the best adsorption capacities towards copper ions.This phenomenon implied that the specific surface area was not the only influencing factor for the adsorption.The adsorption properties of the biochars depended strongly on the type of additive agent used(Table 1 and Fig.1).The adsorption capacities of various modified biochars followed the order 140-8h-1.0-APBC(95.24 mg/g)>140-8h-1.0-TUBC(12.08 mg/g)>140-8h-1.0-ACBC(7.440 mg/g)>140-8h-1.0-URBC(5.277 mg/g).The differences may be due to the different additives agents undergoing different reactions with the raw precursor,resulting in variations in the pore structure and surface chemical properties,especially the surface chemical properties.In the following study,ammonium phosphate was chosen as the activity-enhancing additive due to the excellent adsorption capacity produced with this additive.Ammonium phosphate may be employed as a catalyst to enhance the formation of pores and surface chemical groups during the hydrolysis process by diffusing into the precursor molecules and breaking down the chemical bonds of polysaccharides,hemicelluloses or cellulose compounds.
| 图表编号 | XD0025356200 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.11.15 |
| 作者 | Yuan Gao、Xiuzhen Zhu、Qinyan Yue、Baoyu Gao |
| 绘制单位 | Shandong Provincial Key Laboratory of Water Pollution Control and Resource Reuse,School of Environmental Science and Engineering,Shandong University、Key Laboratory of Industrial Ecology and Environmental Engineering(Ministry of Education),School of Enviro |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1–Langmuir, Freundlich and Dubinin–Radushkevich isotherm parameters for the adsorption of copper ions onto biochar”的人还看了
-

- 表1 Cu O-NPs吸附As (III) 的Langmuir和Freundlich等温吸附模型参数值Tab.1 Langmuir and Freundlich isotherm constants for the As (III) ads
-
- Table 4–Isotherm parameters derived from the Linear, Langmuir, Freundlich, and Dubinin and Radushkevich (D-R) models for
-
- Table 3–Langmuir and Freundlich adsorption isotherm for TMA adsorption by coir pith, lignin and holocellulose extracted
-

- 表1 Langmuir和Freundlich等温吸附模型常数和相关系数Tab.1 Langmuir and Freundlich adsorption isotherm constants andcorrelation coefficien





