《Table 2–Langmuir and Freundlich isotherms fitting parameters for glyphosate adsorption and maximum
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《In situ synthesis of potassium tungstophosphate supported on BEA zeolite and perspective application for pesticide removal》
KF:Freundlich sorption capacity constant;n:Freundlich sorption intensity constant;Sm:adsorbate monolayer;KL:Langmuir sorption constant.
Langmuir and Freundlich adsorption isotherms are often used to model adsorption behavior on the subject of pollutants removal.Fitting results found for these two isotherm models and maximum recorded adsorbed amount of pesticide(qe)are presented in Table 2.Adsorption isotherms for all investigated samples obtained from two models(lines)and experimental adsorption values(squares)are given in Fig.7.The variety of used adsorbents in environmental applications such as soil particles or different composite materials usually have non-homogeneous surfaces,with energetically diverse active sites which involve Freundlich isotherm model for pollutants adsorption.
| 图表编号 | XD0033529600 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.07.15 |
| 作者 | Bojana Nedi Vasiljevi、Milena Obradovi、Danica Bajuk-Bogdanovi、Maja Milojevi-Raki、Zoran Jovanovi、Nemanja Gavrilov、Ivanka Holclajtner-Antunovi |
| 绘制单位 | Faculty of Physical Chemistry, University of Belgrade、Faculty of Physical Chemistry, University of Belgrade、Faculty of Physical Chemistry, University of Belgrade、Faculty of Physical Chemistry, University of Belgrade、Laboratory of Physics, Vina Institute o |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 2–Langmuir and Freundlich isotherms fitting parameters for glyphosate adsorption and maximum recorded adsorbed amo”的人还看了
-
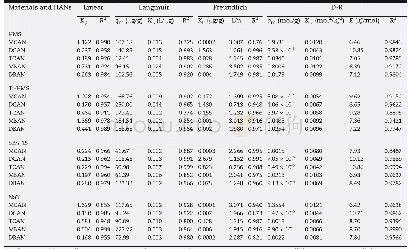
- Table 4–Isotherm parameters derived from the Linear, Langmuir, Freundlich, and Dubinin and Radushkevich (D-R) models for
-
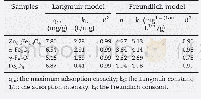
- Table 2–Parameters of Langmuir and Freundlich fits for As (V) adsorption on Zn0.6Fe2.4O4, α-Fe2O3, γ-Fe2O3, and Fe3O4.
-
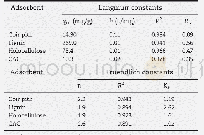
- Table 3–Langmuir and Freundlich adsorption isotherm for TMA adsorption by coir pith, lignin and holocellulose extracted





