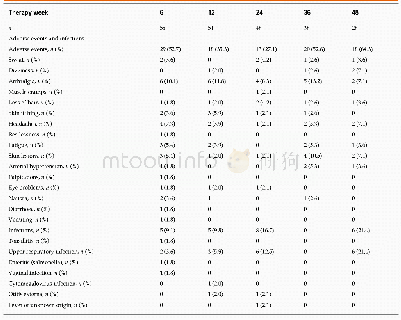
《Table 5 Adverse events during the study treatment》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《定心方联合胺碘酮治疗室性早搏:一项随机、双盲、多中心、安慰剂对照试验(英文)》
Note:*P<0.05 was considered statistically significant.
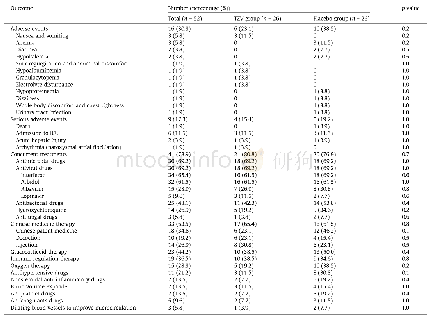
Over 98%of patients in three groups rated the overall tolerability of the three treatments as―good‖or―very good‖.Thirty-six adverse events occurred in 10 participants given amiodarone combined with DXR placebo(13 events),9participants given DXR combined with amiodarone placebo(13 events)and 7 participants given DXR combined with amiodarone(10 events),consisting of 10 cases of rash(4 in the amiodarone group,3 in the DXR group and 3 in the DCA group),9 cases of nausea(3 in the amiodarone group,4 in the DXR group and 2 in the DCA group),7 cases of abdominal pain(3 in the amiodarone group,2 in the DXR group and 2 in the DCA group).Among those,4 events were classified as possibly related to drug.No drug-related serious adverse events occurred during the study treatment,and no statistical difference was observed among the three groups for all adverse events.With regard to laboratory parameters for safety,there were no clinically relevant changes in the three groups(Table 5).
| 图表编号 | XD0014716400 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.03.01 |
| 作者 | 周凤华、贾钰华、叶遂林、娄林洁、杨平、李杰、张月均、钱荣江 |
| 绘制单位 | 南方医科大学、南方医科大学、广州中医药大学、温州大学第一附属医院、江西中医药大学附属中西医结合医院、山西省中医院、横店市人民医院、宁海县中医院 |
| 更多格式 | 高清、无水印(增值服务) |

![Table 3.Cardiovascular Events during 2-Year Clinical Follow-Up[Case (%) ]](http://bookimg.mtoou.info/tubiao/gif/ZXYY201901002_14400.gif)

