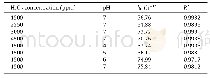
《Table 1 Zero-order rate constants of DNAN oxidation kinetics(k0).》
Assuming that all the H2O2was used to oxidize DNAN to nitrate(HNO3)and CO2,730 ppm H2O2was needed to completely oxidize250 ppm DNAN,as calculated based on Eq.(1).Under experimental conditions,only part of the H2O2was consumed to oxidize the DNAN and its degradation products,and the actual demand for H2O2was greater than 730 ppm.
| 图表编号 | XD00131543100 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.10.01 |
| 作者 | 苏海磊、Christos Christodoulatos、Benjamin Smolinski、Per Arienti、Greg O'Connor、孟晓光 |
| 绘制单位 | Center for Environmental Systems, Stevens Institute of Technology、Center for Environmental Systems, Stevens Institute of Technology、US Army RDECOM-ARDEC、US Army RDECOM-ARDEC、US Army RDECOM-ARDEC、Center for Environmental Systems, Stevens Institute of Techn |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1 Zero-order rate constants of DNAN oxidation kinetics(k0).”的人还看了
-

- 表2 在室温 (298 K) 下团簇Mn3BP各稳定构型转化过程的反应速率常数k和平衡常数K Tab.2 Reaction rate constants (k) and equilibrium constant (K) for change
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式





