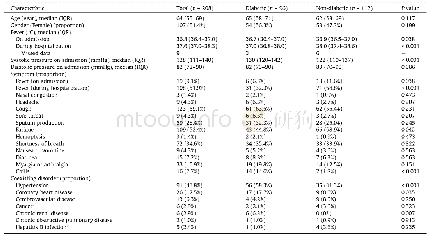
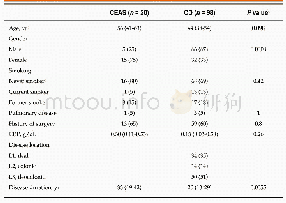
《Table 3 Clinical studies about avelumab, atezolizumab and durvalumab》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《PD-1/PD-L1 pathway blockade works as an effective and practical therapy for cancer immunotherapy》
As shown in Tables 1–3 and many other studies,PD-1/PD-L1 blocking therapies produced a significantly lower rate of high-grade TR adverse events than other immunotherapies,chemotherapies,and standard therapies38,39.This mainly be attributed to the mechanisms of the PD-1/PD-L1 pathway functioning.The PD-1/PD-L1 pathway negatively regulates the immune response mainly in peripheral tissues including the tumor microenvironments40.Moreover,as PD-1/PD-L1blocking therapy mainly activates the inactivated,mature T cells and B cells,and prevents the inactivation of mature T cells and B cells,it mainly affects the late phase of the immune response.Thus,PD-1/PD-L1 blocking therapies produced significantly lower rate of high-grade TR adverse events.According to the studies listed in Tables 1–3,fatigue was the most common TR adverse events.Decreased appetite,asthenia,diarrhea,pneumonitis,rash,and pruritus were also common TR adverse events.
| 图表编号 | XD008421700 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.05.01 |
| 作者 | Long Jia、Qi Zhang、Rongxin Zhang |
| 绘制单位 | Laboratory of Immunology and Inflammation,Department of Immunology and Research Center of Basic Medical Sciences,Key Laboratory of Immune Microenvironments and Diseases of Educational Ministry,Tianjin Medical University、Institute of Integrative Medicines |
| 更多格式 | 高清、无水印(增值服务) |