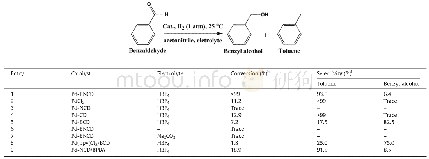
《Table 1 Catalytic hydrogenation of benzaldehyde under mild conditions.a》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Reducing energy barriers of chemical reactions with a nanomicrocell catalyst consisting of integrated active sites in conductive matrices》
aReaction conditions: catalyst(1.5 lmol based on the content of Pd species),benzaldehyde(0.3 mmol),Na2SO4(0.02 mmol)and HBF4(0.08 mmol)or Na2CO3(0.01 mmol)in acetonitrile(1 mL)were stirred at room temperature(25 °C)under an atmosphere of H
Yellow colored boron-and-nitrogen co-doped carbon dots(denoted as BNCD)were synthesized by hydrothermal condensation reaction of a mixture of citric acid(carbon source),disodium edetate(nitrogen source)and p-carboxyphenylboronic acid(boron source).BNCD was subsequently metalated with PdCl2to generate a composite material as a nanomicrocell catalyst(denoted as PdBNCD).In the FT-IR spectrum of BNCD,the weak peak at1,130 cmà1corresponds to the stretching vibration of CAB bonds,and the peak at 1,048 cmà1corresponds to the stretching vibration of CAN bonds,indicating that boron and nitrogen elements were successfully doped in BNCD(Fig.S1 online)[9].The morphology of Pd-BNCD was characterized by transmission electron microscope(TEM)and high-resolution transmission electron microscopy(HRTEM).As shown in Fig.2,the sizes of Pd-BNCD are around 2–5 nm,and the lattice spacing is well matched with that of grapheme[10].There is no aggregated Pd nanoparticle according to the TEM and HRTEM images of Pd-BNCD,indicating that the Pd species was immobilized on BNCD by coordinating to the nitrogen atoms.This conclusion was confirmed by monitoring high-angle annular dark-field scanning transmission electron microscopy(HAADF-STEM)image,energy dispersive X-ray spectroscopy and electron paramagnetic resonance(EPR)spectroscopy(Fig.S2online).The composition of Pd-BNCD was studied by ICP-MS and elemental analysis(Table S1 online).There are about 3.13%Pd,1.08%B and 4.19%N with the Pd,B and N ratio of about 1:3:9 in Pd-BNCD.
| 图表编号 | XD0036052100 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.03.30 |
| 作者 | Guo-Peng Zhan、Chuan-De |
| 绘制单位 | State Key Laboratory of Silicon Materials,Center for Chemistry of High-Performance&Novel Materials,Department of Chemistry,Zhejiang University、State Key Laboratory of Silicon Materials,Center for Chemistry of High-Performance&Novel Materials,Department of |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1 Catalytic hydrogenation of benzaldehyde under mild conditions.a”的人还看了
-
- Table 2 Growth situation of I.polycarpa seedlings under drought stress simulated with different concentrations of PEG





