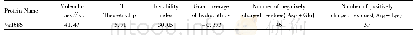《Table 1 Physicochemical properties of the as-prepared catalysts》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Phosphorous-Modified Carbon Nanotube-Supported Pt Nanoparticles for Propane Dehydrogenation Reaction》
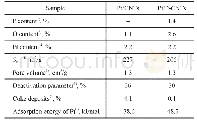
1) Determined by XRF.2) Calculated from the results obtained on an Elementar Vario Micro Cube elemental analyser.3) Estimated from the results of N2 adsorption-desorption isotherms.4) Calculated based on the results in Figure 4.5) Calculated from the resu
Both Pt/P-CNTs and Pt/CNTs catalysts were applied in the PDH reaction,with the corresponding performance exhibited in Figure 4.It can be observed that there is a slight decrease in the propane rate(i.e.,the reaction rate of propane)over Pt/P-CNTs during the PDH reaction,which reduces from the initial value of 124 mmol/(g·h),viz.approximate-ly 2 times more than that over Pt/CNTs(68 mmol/ (g·h)) ,to 87 mmol/(g·h).In comparison,however,the propane rate over Pt/CNTs decreases rapidly from 68 mmol/(g·h)to22 mmol/(g·h).The deactivation parameter(D)is used to measure the catalytic stability,and a low D value suggests a high stability[30].Hence,the catalytic stability over Pt P-CNTs is higher than that over Pt/CNTs(Table 1).Moreover,the propylene selectivity of Pt/P-CNTs is approximately 90%,which is higher by more than 15 percentage points than that of Pt/CNTs(approximately 75%).Generally,the P-modified CNT-supported Pt nanoparticles exhibit superior catalytic activity and stability.
| 图表编号 | XD0029185300 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.03.30 |
| 作者 | Liu Jie、Liu Changcheng、Da Zhijian、Zheng Huidong |
| 绘制单位 | College of Chemical Engineering, Fuzhou University、SINOPEC Research Institute of Petroleum Processing、SINOPEC Research Institute of Petroleum Processing、SINOPEC Research Institute of Petroleum Processing、College of Chemical Engineering, Fuzhou University |
| 更多格式 | 高清、无水印(增值服务) |