《Table 2Initial conditions of the experiments for olivine-CO2-water interaction.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Insight into interactions of olivine-scCO_2-water system at 140C and15 MPa during CO_2 mineral sequestration》
The Specific Surface Area(SSA)of sample powder(75e150 mm)was 0.013 m2/g as determined by BET method.The morphology of the samples were analyzed by a Scanning Electron Microscope and Energy Dispersive X-Ray Spectrometer(SEM-EDS;VEGA II LMUwith BSE detector and Oxford EDS INCA software using 20 keV accelerating voltage;High resolution SEM,FEI Helios NanoLab 650and Oxford EDS AZtec software using 20 keV accelerating voltage).Transmission Electron Microscopy(TEM)were used on a HITACHI H-8100 and TSL-EDAX software with 20 keV accelerating voltage.A D/MAX2500 X-Ray Diffractometer(XRD;Tokyo,Japan)was employed to analyze the compositional change of mineral phases before and after the reaction.The diffraction pattern was scanned over the 2q range of 2.6e45with a scan step of 0.01in continuous mode.The JADEòsoftware with Powder Diffraction File&Inorganic Crystal Structure Database(PDF&ICSD)was used to obtain peak identification and crystallographic information.FT-IR spectra were recorded on a PerkinElmer in the 4000e400 cmà1region,using the KBr disc standard method(2 mg of sample dispersed in 200 mg KBr).Elements(i.e.Mg,Fe,Ca,Na,K,Al,Si)in the reaction solution were determined by Inductively PlasmaOptical Emission Spectrometry(ICP-OES,SPECTRO BLUE SOP).CEC measurements(Chapman,1965)showed the CEC value of sample is 0.4718 meq/L.Briefly,initial mineral(1 g)were extracted using 25 mL aliquots of 1 mol/L ammonium acetate(CH3COONH4).Ammonium hydroxide(NH3$H2O)and acetic acid(CH3COOH)were chosen to keep the solution at pH 7.The initial samples were equilibrated for 15 min by using a table shaker.After that,the samples were centrifuged at 3000 rpm for 15 min.After centrifuging,the supernatant was decanted in a 100 mL volumetric flask.The centrifuging and agitation procedure was repeated four times.The cumulative extract was brought to 100 mL.To remove the suspended solids,the cumulative extracts were then filtered through 0.45 mm pore cellulose acetate filter membranes.The filtrate was acidified by adding three drops of nitric acid(HNO3)and analysis by the ICP-OES.
| 图表编号 | XD0021357200 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.11.01 |
| 作者 | Zijing Li、Jilong Guo、Zhao Dong、Jiawei Chen |
| 绘制单位 | State Key Laboratory of Biogeology and Environmental Geology, China University of Geosciences、School of Earth Sciences and Resources, China University of Geosciences、State Key Laboratory of Biogeology and Environmental Geology, China University of Geoscie |
| 更多格式 | 高清、无水印(增值服务) |
![表2 可靠性实验条件[9]Table 2 Conditions of the reliability experiment[9]](http://bookimg.mtoou.info/tubiao/gif/BDTQ201805003_05200.gif)

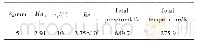

![Table 2 Boundary conditions of experiments in ref.[13]](http://bookimg.mtoou.info/tubiao/gif/TJJS201901009_08300.gif)