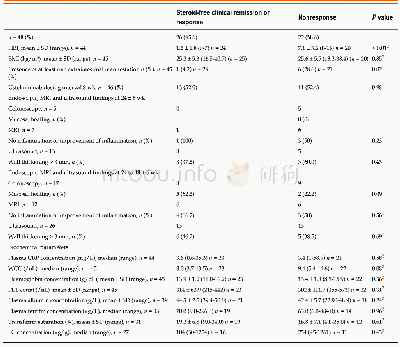
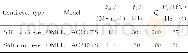《Table 5 Comparison between parameters determined at 24±6 wk from start of ustekinumab therapy betwe
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Ustekinumab: “Real-world” outcomes and potential predictors of nonresponse in treatment-refractory Crohn's disease》
In conclusion,our data strongly suggest that ustekinumab is effective in treatmentrefractory,moderate to severe CD under“real-world”conditions.In particular,more data is needed on the long-term safety of this drug and on potential predictors of response.
| 图表编号 | XD0087253400 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.08.21 |
| 作者 | Peter Hoffmann、Johannes Krisam、Cyrill Wehling、Petra Kloeters-Plachky、Yvonne Leopold、Nina Belling、Annika Gauss |
| 绘制单位 | Department of Gastroenterology and Hepatology, University Hospital Heidelberg、Department of Medical Biometry, University Hospital Heidelberg、Department of Gastroenterology and Hepatology, University Hospital Heidelberg、Department of Gastroenterology and H |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 5 Comparison between parameters determined at 24±6 wk from start of ustekinumab therapy between steroid-free clini”的人还看了
-
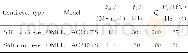
- Table 1 Comparison of the essential parameters between the commercial soft and stiff cantilever AFM probes produced by O
-
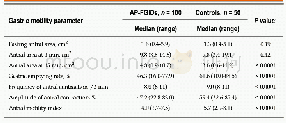
- Table 3 Comparison of gastric motility parameters between children with abdominal pain-predominant functional gastrointe
-

- Table 2 Comparison of autonomic parameters between children with abdominal pain-predominant functional gastrointestinal