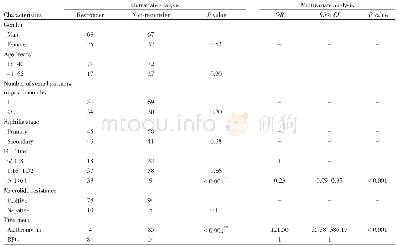
《Table 3 TB risk in Chinese patients treated with biologic agents》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Biologic treatments for psoriasis in China: current status and potential tuberculosis and hepatitis B virus risks》
RCT:randomized controlled trial;RS:retrospective study;PS:prospective study;OLS:Open-label study;UC:ulcerative colitis;AS,:ankylosingspondyltis;RA:rheumatoid arthritis;JIA:juvenile idiopathic arthritis;PsA:psoriatic arthritis;TB:tuberculosis;NR:not report
Current data in China demonstrate that Chinese patients treated with biologics are at greater risk for developing TB than Western populations(Table 3).During the whole 26-week study period in the Phase III trials for infliximab,a total of 3 cases of active TB were reported as severe adverse events in 84 psoriasis patients[13].The incidence rate was3.5%,and higher than the results in the trials from US[20],Europe[21],and Japan[22].TB occurred 11 to 26weeks after the initiation of infliximab treatment.Among the cases,2 were reported to be pulmonary TB,while another case was reported to be TB pleurisy.In a single-center,retrospective study,1case of severe TB was identified in 24 ulcerative colitis Chinese treated with infliximab,leading to discontinuation of infliximab therapy[23].
| 图表编号 | XD00196121800 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.06.01 |
| 作者 | Meng Fu、Gang Wang |
| 绘制单位 | Department of Dermatology, Xijing Hospital, The Fourth Military Medical University、Department of Dermatology, Xijing Hospital, The Fourth Military Medical University |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 3 TB risk in Chinese patients treated with biologic agents”的人还看了
-

- Table 5 Multivariable analysis of risk factors for in-hospital mortality in patients with hepatic cirrhosis
-

- Table 4 Multivariable analysis of risk factors for development of a major bleed in patients with hepatic cirrhosis