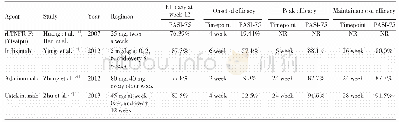
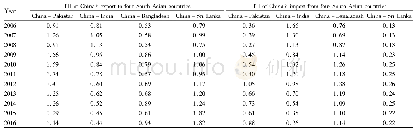
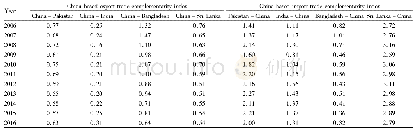
《Table 1 Basic properties of the four biologics in China》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Biologic treatments for psoriasis in China: current status and potential tuberculosis and hepatitis B virus risks》
*Phase III clinical trials in psoriasis have been completed,and the biologics will be approved by China’s FDA for the treatment of psoriasis soon.
The efficacies of the four biologic agents in treatment of moderate and severe psoriasis in China are described in Table 2.A Phase III study compared effects of Yisaipu(50 mg)once weekly combined with methotrexate(MTX)for 12 weeks on 144 patients with moderate or severe psoriasis vulgaris[11-12].At week 12,the researchers found that 76.39%of patients treated with Yisaipu achieved PASI-75,compared with 44.44%of patients,who received MTX(P<0.01).Significant differences were recorded between the two groups as early as week 4 forthe proportion of patients achieving PASI 75.At this time point,19.44%of the patients treated with Yisaipu achieved PASI-75,compared with 4.17%of patients who received MTX(P<0.01).
| 图表编号 | XD00196121500 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.06.01 |
| 作者 | Meng Fu、Gang Wang |
| 绘制单位 | Department of Dermatology, Xijing Hospital, The Fourth Military Medical University、Department of Dermatology, Xijing Hospital, The Fourth Military Medical University |
| 更多格式 | 高清、无水印(增值服务) |