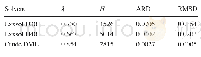《Table 2 Parameters obtained from the polarization curves in Fig.3》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《pH值对镁合金表面水热沉积磷酸钙涂层微观形貌和腐蚀性能的影响(英文)》
The polarization curves of specimens in Hank's solution are shown in Fig.3.The electrochemical parameters fitted from the polarization curves are listed in Table 2.The polarization curves of all the coated samples display distinct passivation regions.Although the Ecorr(corrosion potential)values of the hydrothermally treated specimens are lower than that of the AZ31 substrate,the Ebk(breakdown potential)is much higher than that of the AZ31 substrate,which indicates that the hydrothermally deposited coatings provide effective protection for magnesium alloys.Table 2 shows that the icoor(corrosion current density)of the pH=6-treated specimen is very close to that of the AZ31 substrate.While the icoor values of the pH=8 and pH=10-treated specimens are nearly 10 times lower than that of the AZ31 substrate.Among the hydrothermally treated specimens,the OCP coated specimen treated at pH=6 has the highest Ebk,indicating that the formed coating protects the substrate more effectively than the other two coated specimens.However,icoor of OCP coated specimens is the highest,which suggests that when the coating is broken,the corrosion rate will be faster than that of other coated specimens.The Ebk of pH=10-treated specimen is close to that of the pH=8-treated specimen,but the icoor is slightly lower.
| 图表编号 | XD0011274000 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.10.01 |
| 作者 | 张春艳、张世雨、柳歆鹏、何洪川 |
| 绘制单位 | 重庆理工大学、重庆市特种焊接材料与技术高校工程研究中心、重庆理工大学、重庆理工大学、重庆理工大学 |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 2 Parameters obtained from the polarization curves in Fig.3”的人还看了
-

- 表3 Ni-Ti N纳米镀层的Tafel曲线拟合参数Table 3 Fitting parameters obtained from Tafel curves of Ni-Ti N nanocoatings
-

- Table 2 Distribution of changes in time-intensity curve parameters in tumors from day 0 to day 7for 20 hepatocellular ca
-
- Table 3 Curve fi tting parameters of theλh equation for fl uorene in Exxsol D30, Exxsol D40, and crude DME from 299.25 t
-

- Table 4 Adsorption parameters obtained from mass loss measurements for the studied compound in 1 mol/L HCl at different