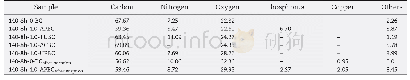
《Table 1Gel compositions and resultant products of hydrothermal syntheses.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《"以N,N’-二甲基乙二胺为模板剂合成磷酸硅铝分子筛DNL-6及其在催化中的应用(英文)"》
a The crystallization conditions are 200°C and 12h.b The crystallization time is 68 h for SP-D4 and 143 h for SP-D5.
The detailed synthesis conditions including gel composition,crystallization time,product crystalline phases,and composition are listed in Table 1.The corresponding XRD patterns of these products are shown in Fig.1.When TEOS is absent(SP-D1),Al PO4-21 with an AWO topology is the final product.The AWO phase decreases and RHO phase increases with anincrease in the amount of TEOS.Pure DNL-6 molecular sieves cannot be synthesized unless the molar ratio of TEOS/Al2O3 is1.Further,an amorphous phase appears along with DNL-6when the TEOS/Al2O3 molar ratio is greater than 1.7.Pure-phase DNL-6s synthesized at TEOS/Al2O3 ratios of 1.0,1.2,and 1.7 were denoted as L-DNL-6,M-DNL-6,and H-DNL-6,respectively.Their Si contents range from 22.4%to 36.4%.It should be noted that SAPO molecular sieves with such high Si contents are rarely reported.Upon further prolonging the crystallization time,the RHO phase gradually converts to a dense GIS phase(68 h for SP-D4 and 143 h for SP-D5),indicating the metastable characteristics of DNL-6.Meanwhile,the Si content of solid products is higher than that of M-DNL-6.
| 图表编号 | XD008196300 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.09.01 |
| 作者 | 吴鹏飞、杨淼、张雯娜、曾姝、高铭滨、徐舒涛、田鹏、刘中民 |
| 绘制单位 | 中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大学、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大学、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大学、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大学、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室、中国科学院大连化学物理研究所甲醇制烯烃国家工程实验室 |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1Gel compositions and resultant products of hydrothermal syntheses.”的人还看了
-

- Table 4 Product composition of diff erent adsorbent used model fuel:1-hexene (34.3%) and n-heptane (65.5%)