《Table 3 Results of accuracy and precision of the developed RP-HPLC method using MAC and its associa
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《指示稳定性的高效液相色谱方法和强制降解研究片剂型马西替坦中的杂质(英文)》
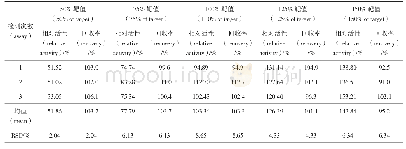
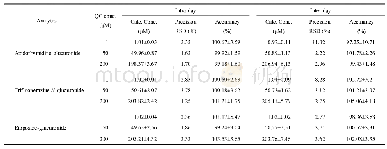
Accuracy o f the develo ped m etho d is determ ined by m easuring the am o unt o f reco vered sam ples by injecting o r spiking kno w n co ncentratio ns o f im purities in the pure drug sam ple.The co ncentratio ns cho sen fo r accuracy determ inatio n w ere LOQ,100%,and 150%,specificatio n lim it as 0.5%(m ass percentage),w ith three replicate m easurem ents.The chro m ato gram s fo r the spiked sam ples fo r accuracy determ inatio n w ere sho w n in Fig.8 and the results are sum m arized in Table3.Results sho w ed that the develo ped m etho d can successfully reco ver the injected im purities w ithin the range o f 85%-115%,w hich is m o re accurate than o ther repo rted m etho do lo gies[33,34].
| 图表编号 | XD0041365300 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.01.08 |
| 作者 | Narasimha S LAKKA、Chandrasekar KUPPAN、Parthasarathy RANGASAMY |
| 绘制单位 | Department of Science and Humanities,VIGNAN's Foundation for Science,Technology & Research (VFSTR)、Department of Analytical Research & Development,Sinotherapeutics Inc.、Department of Science and Humanities,VIGNAN's Foundation for Science,Technology & Re |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 3 Results of accuracy and precision of the developed RP-HPLC method using MAC and its associated impurities in the”的人还看了
-

- Table 6Recall,precision,f1-score,and accuracy rateaof the export of the Noisy-OR Bayesian function for COVID-19,IAVP,and