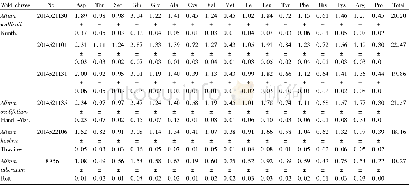
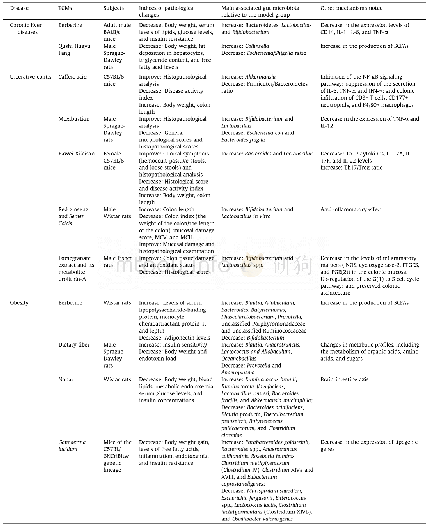
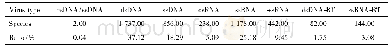《Table 1Pharmacopoeia and directives of TCMs in different countries or regions.》
In recent years,the need for a TCM quality control system has become a new hotspot attracting a great deal of research interest.A few years ago,the new concept of a TCM quality marker was proposed by Prof.Changxiao Liu et al.[14,15].The present review discusses new methods and technologies to explore potential quality markers for TCM quality control,considering the complexity of TCM and the factors that affect TCM quality,and may provide insight into rational grounds for the establishment of an appropriate TCM quality control and assessment system.This paper also discusses progress that has been made in TCM quality control,with the aim of providing a valuable reference for future investigations.
| 图表编号 | XD00131543800 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.10.01 |
| 作者 | 李亚卓、谢莹、赫宇霏、侯文彬、廖茂梁、刘昌孝 |
| 绘制单位 | State Key Laboratory of Drug Delivery Technology and Pharmacokinetics, Tianjin Institute of Pharmaceutical Research、State Key Laboratory of Quality Research of Chinese Medicines, Macau University of Science and Technology、School of Pharmacy, Shenyang Phar |
| 更多格式 | 高清、无水印(增值服务) |
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式