《Table 2–Trimethylamine (TMA) adsorption capacity of various adsorbents.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Lignin and holocellulose from coir pith involved in trimethylamine(fishy odor) adsorption》
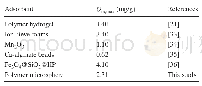
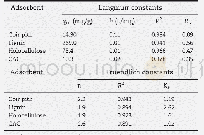
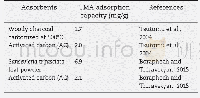
For soluble TMA in oil,H-type faujasite zeolite and H-type mordenite zeolite were reported as effective adsorbents to adsorb soluble TMA in fish oil,which presented 87.7 and63.9 mg/g of TMA adsorption capacity,respectively(Chung and Lee,2009).For gaseous TMA,various adsorbents including S.trifasciata and P.longifolia leaf materials were reported as effective gaseous TMA adsorbents,which presented to harbor a higher gaseous TMA adsorption capacity than CAC(Chung and Lee,2009;Boraphech and Thiravetyan,2015).Since Boraphech and Thiravetyan(2015)proposed that lignin in S.trifasciata and P.longifolia leaf materials played the role to adsorb TMA,high lignin materials might be effective adsorbents for TMA adsorption.Coir pith contained higher lignin levels(36.0%)than S.trifasciata(5.8%)and P.longifolia leaf(8.7%)(Suksabye et al.,2009;Sathishkumar et al.,2014;Ogunbosoye et al.,2015) .Therefore,coir pith can probably be an effective gaseous TMA adsorbent.This study,TMAadsorption by S.trifasciata leaf material,P.longifolia leaf material,CAC and coir pith were compared.The result showed that coir pith was an effective material for TMA,which adsorbed 93.2%of 100 ppmV TMA within 24 hr.The adsorption efficiency of coir pith was significantly higher than other materials,while CAC normally had the ability to remove the odors and widely used for waste gas treatments in several industries(Tsutomu et al.,2004;Lee et al.,2010),could not well adsorb TMA(Fig.3).The result can be explained by the characteristic of CAC which was not suitable to adsorb gaseous TMA.Gaseous TMA is a polar molecule,while CACwas mostly used for non-polar gaseous pollutants(Ren et al.,1987;Tsutomu et al.,2004;Lee et al.,2010).In summary,coir pith had the highest TMA adsorption capacity among all materials from the screening(Table 2).This might be due to the high lignin composition of coir pith.Pseudo-first and second order kinetics and their parameters for TMA adsorption by using 0.1 g of coir pith under 100 ppmV of TMA concentration were calculated.The calculated adsorption capacity at equilibrium(qe,cal)from pseudo-first order was 24.5 mg/g and qe,calfrom pseudo-second order was 33.3 mg/g,while the experimental adsorption capacity at equilibrium(qe,exp)was35.1 mg/g.Therefore,pseudo-second order was more fitted than pseudo-first order.
| 图表编号 | XD0052052600 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.05.15 |
| 作者 | Nuttapong Santawee、Chairat Treesubsuntorn、Paitip Thiravetyan |
| 绘制单位 | School of Bioresources and Technology, King Mongkut's University of Technology Thonburi、Pilot Plant Development and Training Institute, King Mongkut's University of Technology Thonburi、School of Bioresources and Technology, King Mongkut's University of |
| 更多格式 | 高清、无水印(增值服务) |