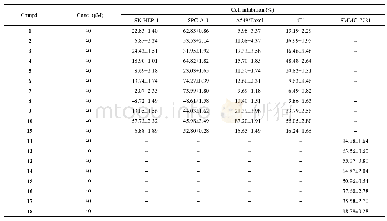
《Table 1 Inhibitory activities, free energy of binding, H-bond interactions, and hydrophobic interac
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《"Protein tyrosine phosphatase 1B inhibitory activities of ursane-type triterpenes from Chinese raspberry, fruits of Rubus chingii"》
a IC50 were determined by regression analyses and expressed as mean±SD of three replicates;b IC50 from reference[23].
Triterpenes 1-3 were evaluated PTP1B inhibitory activities at different concentrations.As a result,1-3 inhibited PTP1B in a concentration dependent manner,and the IC50were determined by regression analyses(Table 1).The inhibition modes of compounds 1-3 were further elucidated by kinetics analyses with various concentrations of the compounds and the substrate p-nitrophenyl phosphate(p-NPP).As shown in Fig.2,Lineweaver-Burk plots indicated compounds 1 was a non-competitive PTP1B inhibitors with the Ki of 25.8μmol·L-1,and compounds 2 and 3 inhibited PTP1B by a mixed mode.The secondary plots of compounds 1 and 2showed good linear relationship,when they were created by the slopes from the Lineweaver-Burk plots on the y-axis against the concentration on the x-axis.Meanwhile,the secondary plot of compound 3 showed good linear relationship,when plotting[I]2 on the x-axis,suggesting that two molecules of 3 were involved in the inhibition of PTP1B activity.
| 图表编号 | XD0047580600 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.01.20 |
| 作者 | ZHANG Xiang-Yu、LI Wei、WANG Jian、LI Ning、CHENG Mao-Sheng、KOIKE Kazuo |
| 绘制单位 | Key Laboratory of Structure-Based Drug Design & Discovery,Ministry of Education,Shenyang Pharmaceutical University、School of Pharmaceutical Engineering,Shenyang Pharmaceutical University、Key Laboratory of Structure-Based Drug Design & Discovery,Ministry o |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1 Inhibitory activities, free energy of binding, H-bond interactions, and hydrophobic interactions between triterp”的人还看了
-
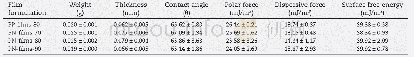
- Table 2–Weight, thickness, contact angle, polar force, dispersive force and surface free energy of PP-films and PN-films