《Table 1 Tentative assignment of glycinin in Raman spectra》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Study on Isolation and Raman Spectroscopy of Glycinin in Soybean Protein》
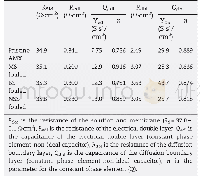
The doublet at 850 and 830 cm-1in the Raman spectra of proteins containing tyrosyl residues was examined to determine its origin and detect Fermi resonance between the symmetric ring-breathing vibration and overtone of the out-of-plane vibration of the para-substituted benzene ring.This information could also reflect whether the tyrosine residues of proteins were exposed or buried[7].When I850/I830 was 1.25–1.40,0.7,and 0.3–0.5,the residues representing tyrosine were completely exposed,ionized,and embedded in the surface of proteins molecules,respectively[20].In this study,the characteristic band of tyrosine side chains in glycinin was I850/I830=1.145 at 642,834,855,and 1 204 cm-1.Furthermore,in accordance with the N(buried or exposed)calculation method described by Kalapathy[20],the N-buried and N-exposed tyrosine residues account for 14.1%and 85.9%of the total amount of residues in glycinin,respectively(Table 3).Thus,the tyrosine residues evaluated in this study tended to be exposed.
| 图表编号 | XD0021321400 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.04.30 |
| 作者 | YIN Haicheng、HUANG Jin、ZHANG Huiru |
| 绘制单位 | College of Biological Engineering, Henan University of Technology、Grain & Corn Engineering Technology Research Center,State Administration of Grain、College of Biological Engineering, Henan University of Technology、College of Biological Engineering, Henan |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1 Tentative assignment of glycinin in Raman spectra”的人还看了
-
- Table 3–Fitting results of the impedance spectra shown in Fig.8 obtained in the AEM systems according to the equivalent
-

- 表2 γ-Na2TiO3晶型晶体主要拉曼活性振动模式归属Table 2 The assignment of the major Raman-active vibrational modes forγ-Na2TiO3crystal
-

- 表4 图7中27 Al NMR图谱去卷积计算结果Table 4The calculation results from deconvolution of 27 Al NMR spectra in Fig 7





