《Table 1.Kinetic parameters for the leaching reaction of Li (pH 2, 2vol%H2O2, and an S/L ratio of 10
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Reaction kinetics modeling for lithium and cobalt recovery from spent lithium-ion batteries using acetic acid》
Note:Forward reaction order=2,backward reaction order=1.
The data processing results in Figs.3 and 4 indicate that the calculation results do not substantially deviate from the data obtained from the experiments,which means that the kinetics model sufficiently represents the reaction kinetics in the leaching experiments.The results of the calculations of the kinetics parameters for leaching of Li and Co are summarized in Tables 1 and 2.
| 图表编号 | XD0028410100 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2019.01.01 |
| 作者 | Hendrik Setiawan、Himawan Tri Bayu Murti Petrus、Indra Perdana |
| 绘制单位 | Sustainable Mineral Processing Research Group, Department of Chemical Engineering, Faculty of Engineering, Universitas Gadjah Mada、Sustainable Mineral Processing Research Group, Department of Chemical Engineering, Faculty of Engineering, Universitas Gadja |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 1.Kinetic parameters for the leaching reaction of Li (pH 2, 2vol%H2O2, and an S/L ratio of 10 g/L)”的人还看了
-

- Table 2.Total number of patients administrated of IVIG and the incidence of adverse reactions for 3 consecutive years.
-
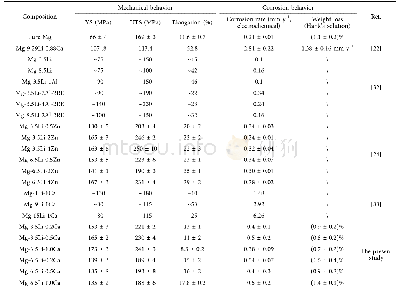
- Table 4 Summary of the mechanical and corrosion behavior of reported Mg-Li based alloys for biomedical application





