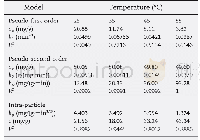
《Table 4 Main reactions and kinetic parameters in the char combustion and gasification.》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Effects of High-temperature Char Layer and Pyrolysis Gas on NOx Reduction in a Typical Decoupling Combustion Coal-fired Stove》
HCN and NH3 are the main light nitrogen species produced by the pyrolysis of coal,and the ratio between the two species is in close relation to fuel type,pyrolysis temperature and heating rate[59-61].At high heating rates and small particle sizes,HCN is generally the dominant light nitrogen in the devolatilization of bituminous coal[60,62].In this study,the volatile N is assumed to be completely transformed into HCN and NH3 under the conditions of low temperature and reduction atmosphere,and HCN/NH3 partition mole ratio of 7/3 is used as a preliminary test[63,64].The transformation rate of the char N is generally 40%,about80%of which is trans-formed to NO and the residual to HCN and NH3 at the mole ratio HCN/NH3=7/3 too[63-66].Thereby,the transformation route of fuel N is summarized in Fig.2.The relevant reactions including the reduction of NO to N2 are listed as well as corresponding kinetics parameters in Table 5[67-69].
| 图表编号 | XD0039671300 严禁用于非法目的 |
|---|---|
| 绘制时间 | |
| 作者 | LI Honglin、HAN Jian、ZHANG Nan、LIU Xinhua、HE Jingdong、DU Wei |
| 绘制单位 | State Key Laboratory of Multiphase Complex Systems, Institute of Process Engineering, Chinese Academy of Sciences、State Key Laboratory of Heavy Oil Processing, China University of Petroleum、State Key Laboratory of Multiphase Complex Systems, Institute of |
| 更多格式 | 高清、无水印(增值服务) |