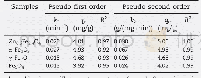
《Table 2 Parameters of linear fitting of adsorption equilibrium》
 提示:宽带有限、当前游客访问压缩模式
提示:宽带有限、当前游客访问压缩模式
本系列图表出处文件名:随高清版一同展现
《Preparation and SO_2 Adsorption Behavior of Coconut Shell——Based Activated Carbon via Microwave-Assisted Oxidant Activation》
As shown in Table 2,the results of the Langmuir adsorption isotherm model are better than those of the Freundlich model,which means that the Langmuir model can better predict the adsorption equilibrium of SO2 on the surfaces of the samples.According to this model,the samples mainly demonstrate the chemical surface adsorption behavior for SO2.
| 图表编号 | XD0011296000 严禁用于非法目的 |
|---|---|
| 绘制时间 | 2018.03.30 |
| 作者 | Jia Fengrui、Li Zhou、Wang Engang、He Jicheng、Dong Hui、Liu Guangxin、Jian Weiwei |
| 绘制单位 | School of Metallurgy, Northeastern University、College of Petroleum Engineering, Liaoning Shihua University、College of Petroleum Engineering, Liaoning Shihua University、School of Metallurgy, Northeastern University、School of Metallurgy, Northeastern Univer |
| 更多格式 | 高清、无水印(增值服务) |
查看“Table 2 Parameters of linear fitting of adsorption equilibrium”的人还看了
-
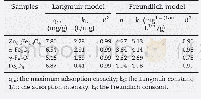
- Table 2–Parameters of Langmuir and Freundlich fits for As (V) adsorption on Zn0.6Fe2.4O4, α-Fe2O3, γ-Fe2O3, and Fe3O4.